Clean room
Our Clean Room has 6 injection machines of up to 160 Tm, a screen printing machine to decorate dosing syringes and vaginal applicators and a bagger in a PP film flow pack.
In addition to the manufacture and decoration of the pieces, we also have an assembly and handling area for medical devices.
Environmental Control of ISO 7 Clean Room
The existence of overpressure inside the manufacturing area towards the exchangers (SAS Material and SAS Personal), and of these with respect to the outside, prevents the entry of air that has not circulated through the ultrafiltration system.
The protocol for personnel access to the manufacturing area includes the use of clothing made of specific materials that contribute to low particle emissions.
Our Clean Room has the ISO 7 / Class C environmental qualification according to the validation protocol carried out in compliance with the ISO 14698 standard on an annual basis.

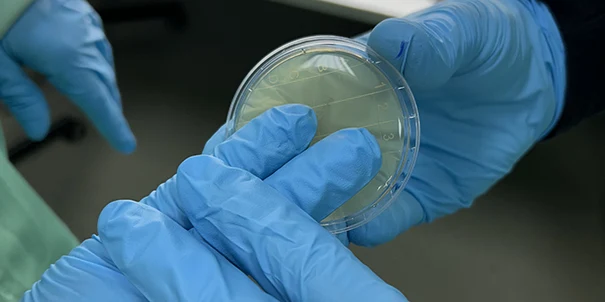
Microbiological control of the Clean Room
It is a guarantee that the filtration system is working correctly and the manufacturing environment is safe.

Clean Room Cleaning and Disinfection Protocol
The disinfection of work surfaces is validated by microbiological cultures from sampling with contact plates.

Highly qualified staff
All Triana staff are involved in continuous training projects. In this way, by acting on the different critical points of the process according to the risk assessment and analysis carried out, we close the cycle of quality assurance of the manufactured product.
Designed and made in Barcelona

