Medical devices
We are legal manufacturers of medical devices, and also manufacturers for third parties. We have an ISO 7 Clean Room and are ISO 13485 certified, ensuring compliance with international regulations and safety in clinical use.
Types of medical devices
Medical devices are defined as any instrument, apparatus, equipment, software, material or other article, used alone or in combination, including software, intended by the manufacturer to be used for the purposes of diagnosis and/or therapy and which are involved in its proper functioning, intended by the manufacturer to be used in human beings for the purposes of:
- Diagnosing, preventing, monitoring, treating or alleviating a disease.
- Diagnosing, monitoring, treating, alleviating or compensating for an injury or impairment.
- Investigating, replacing or modifying the anatomy or a physiological process.
- Regulation of conception.
And which does not exert the principal intended action in or on the human body by pharmacological, immunological or metabolic means, but which may be assisted in its function by such means.
What makes us unique in the manufacture of medical devices

Clean Room and Dry Room Production
We manufacture all our medical devices in controlled Clean Room and Dry Room environments, where air purity, temperature and humidity are strictly regulated. This process guarantees the absence of contaminating particles and maximum hygiene during production and packaging.
The combination of both rooms allows us to maintain optimal conditions for sensitive materials and products that require protection from humidity. This environmental control ensures the stability, functionality and sterility of every medical component we manufacture.
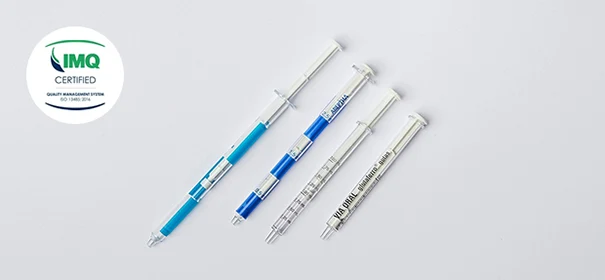
ISO 13485 Certification for Medical Devices
Our production is backed by ISO 13485 certification, which accredits compliance with the highest international standards in quality management systems for medical devices.
This certification guarantees complete product traceability, process validation and strict regulatory compliance at all stages of design, manufacture and control. It is a demonstration of our commitment to the safety and reliability of the medical devices we offer.

CE Marking for Medical Devices
Our products carry the CE marking, which certifies their compliance with the essential safety, quality and performance requirements established by European regulations for medical devices. This accreditation guarantees that each manufactured component has passed the necessary technical and documentary checks prior to being placed on the market.
Compliance with Directive 93/42/EEC and Regulation (EU) 2017/745 (MDR) reinforces the reliability of our processes and the safety of use of our devices. Thanks to this certification, we offer our customers the peace of mind of working with products that meet the most demanding standards of the European market in the field of healthcare.
of experience in packaging
in ISO 7 Cleanroom and Dry Room
parts manufactured
per year
Certificate for
Medical Devices
Pharmacy and
Parapharmacy
Designed and made in Barcelona